12 Modules. Zero Governance Gaps.
Each module addresses a distinct dimension of nutraceutical governance failure. Together, they constitute a complete operating system — from the first adverse signal to the final documented action.
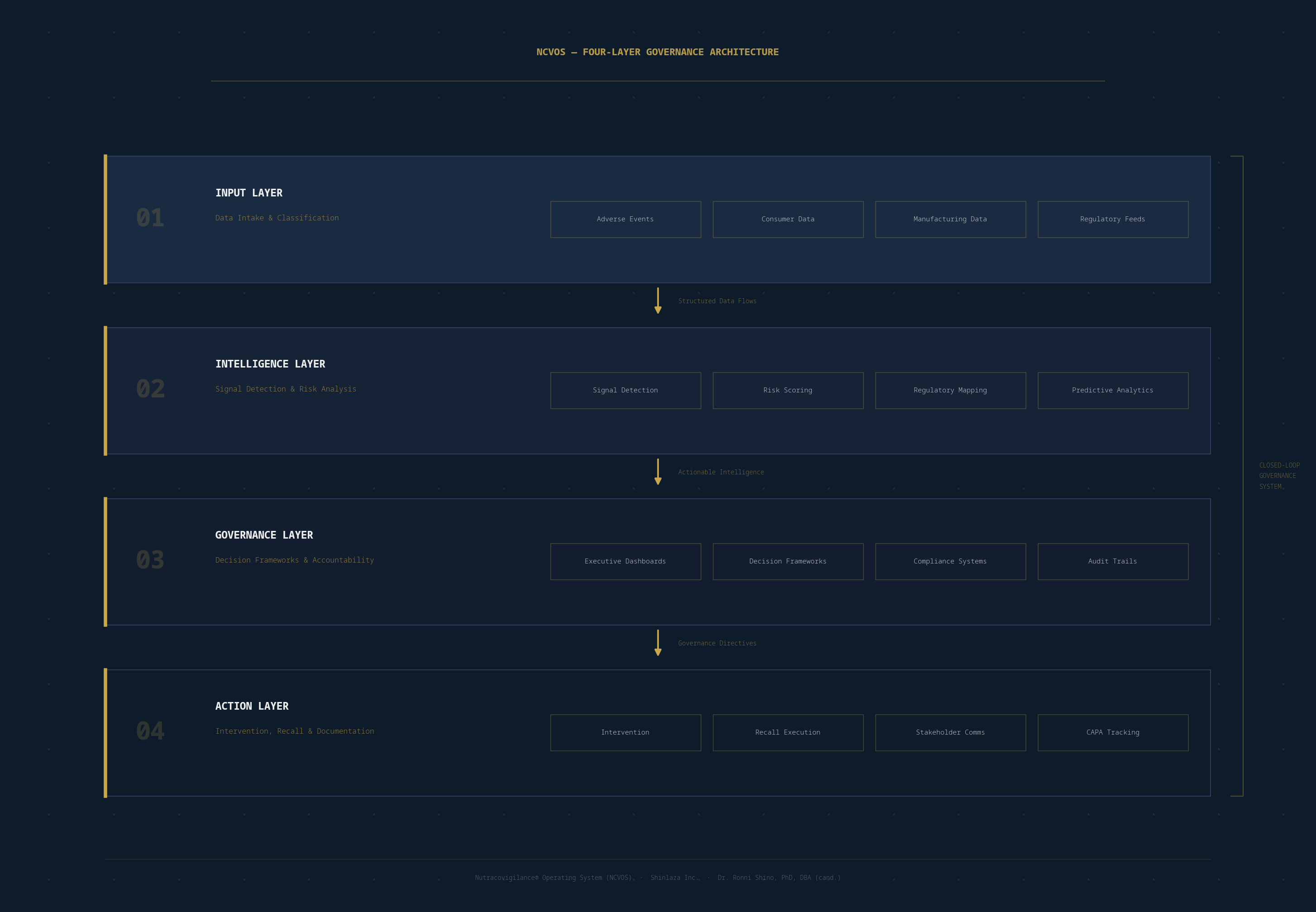
The diagram below maps the complete four-layer governance flow of NCVOS — from structured data intake through intelligence processing, governance decision-making, and documented intervention. Each layer is interdependent; the system functions as a closed loop with no accountability gaps.
NCVOS Four-Layer Architecture · Shinlaza Inc. · Dr. Ronni Shino, PhD, DBA (cand.)
Request Pilot Access
NCVOS is entering its institutional pilot phase on May 1, 2026. Following successful internal testing, the platform is now accepting structured expressions of interest from governments, regulatory bodies, enterprises, and institutions seeking to adopt Nutracovigilance® as operational infrastructure.
Pilot access is limited and subject to jurisdictional alignment review. Priority consideration is given to government bodies and regulatory agencies in active engagement jurisdictions.
Direct Contact
Compliance & Regulatory
[email protected]Partnerships & General
[email protected]Research & Academic
[email protected]Pilot Access Inquiry Form